47% Of Women Who Exercise Regularly Experience Stress Incontinence1
What Is Stress Urinary Incontinence (SUI)?
Stress urinary incontinence is the unintentional leaking of urine upon exertion. That can be sneezing, coughing, jumping, running or other physical activities. If you are leaking a few drops of urine when coughing or sneezing this is a sign of stress incontinence. Stress incontinence can also happen when participating in high impact sports or lifting heavy weights. For example; running, dancing, horse riding, trampolining, gymnastics, cross-fit and weight lifting are all activities that can highlight stress incontinence symptoms. If you are leaking with less forceful activities, like standing up, walking or bending over, this indicates your incontinence is more severe.
Causes of Stress Urinary Incontinence (SUI)
Stress urinary incontinence is generally the result of weakness or injury to the pelvic floor muscles. It can be caused by pregnancy or childbirth. Other factors such as age, BMI, smoking, high impact sports and previous pelvic surgeries can contribute to stress incontinence. Stress incontinence is common following menopause, which is believe to be due to hormonal changes.
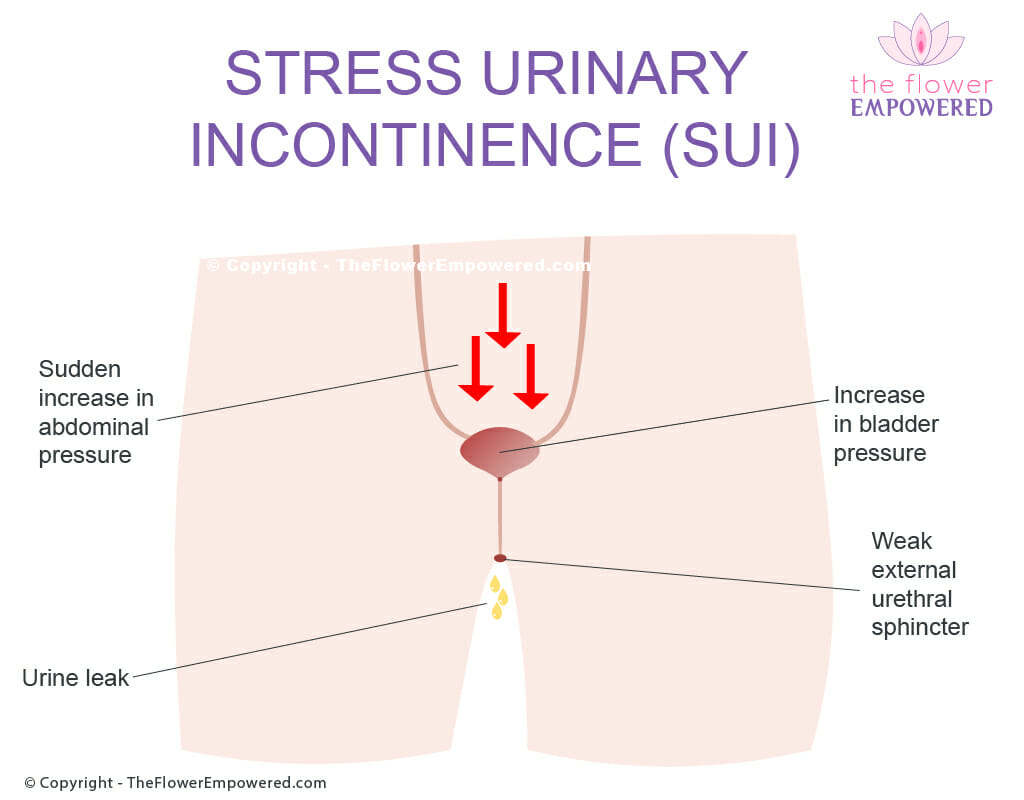
Stress Urinary Incontinence Explained
We’ve created the image to show what happens when you have an episode of stress incontinence.
A sudden increase in abdominal pressure from exertion (cough, sneeze, laugh, jumping, lifting something heavy etc.) causes an increase in intraabdominal pressure.
The increased intra-abdominal pressure causes an increase in bladder pressure.
If the external urethral sphincter cannot match the increase in pressure with an increased pressure of contraction, leakage occurs.
Once the increase in intraabdominal pressure has passed, bladder pressure returns to normal. The external urethral sphincter can hold in urine with normal bladder pressure.
If you are experiencing stress incontinence, even if it’s just a small amount, this is an indication that you may have some weakness in your external urethral sphincter which is a muscle in your pelvic floor. Strengthening your pelvic floor can help to ensure that you regain control over this muscle and prevent future leakages. Even if your stress incontinence is severe, you can still make big improvements. Studies show that women who have weaker pelvic floor muscles to begin with have the greatest percentage improvement in symptoms from pelvic floor muscle training. You can read more about pelvic floor muscle training when reviewing the conservative treatments for urinary incontinence.
Stress Urinary Incontinence Is the Most Prevalent Type of Incontinence in Women2
Products for Management of Stress Urinary Incontinence
There are many products that can be used in the management of pelvic organ prolapse. We have include some of these products below. You should speak with your pelvic floor physiotherapist or doctor before using these products. Reviews of these products will be covered on our YouTube channel. If there is a specific product that you would like to see reviewed, just get in touch and we will review. Note that these products have affiliate links. Should you choose to purchase them, we will make a small commission.
Treatment Options for Urinary Incontinence
Like most symptoms of pelvic floor dysfunction, the treatment for urinary incontinence falls into one of two categories; conservative (non-surgical) and invasive (surgical). Surgical options are generally only advised for once conservative options have been tried unsuccessfully for an extended period. For more information on the various treatments offered in these two categories, click on the relevant button below. If you did not find what you were looking for, you can search this site using the search bar at the bottom of the page.
Explore the Other Types of Urinary Incontinence
Having reviewed the Mixed Urinary Incontinence, you may be interested in exploring the other types of incontinence. You can do that from here:
References
- Pires T, Pires P, Moreira H, Viana R. Prevalence of Urinary Incontinence in High-Impact Sport Athletes: A Systematic Review and Meta-Analysis. J Hum Kinet. 2020 Jul 21;73:279-288. doi: 10.2478/hukin-2020-0008. PMID: 32774559; PMCID: PMC7386138.
- Moossdorff-Steinhauser HFA, Berghmans BCM, Spaanderman MEA, Bols EMJ. Prevalence, incidence and bothersomeness of urinary incontinence in pregnancy: a systematic review and meta-analysis. Int Urogynecol J. 2021 Jul;32(7):1633-1652. doi: 10.1007/s00192-020-04636-3. Epub 2021 Jan 13. PMID: 33439277; PMCID: PMC8295103..